
Therefore, they often vary since one is theoretical and the other is measured. It should be noted, however, that the molar mass is almost always a computed figure derived from the standard atomic weights, whereas the average molecular mass, in fields that need the term, is often a measured figure specific to a sample. The average molecular mass and the molar mass of a particular substance in a particular sample are in fact numerically identical and may be interconverted by avogadro's number. This is often closer to what is meant when "molecular mass" and "molar mass" are used synonymously and may have derived from shortening of this term. The average molecular mass is the abundance weighted mean (average) of the molecular masses in a sample. The average molecular mass (sometimes abbreviated as average mass) is another variation on the use of the term molecular mass. The actual difference numerically is very small and only matters to physicists and a small subset of highly specialized chemists however it is always more correct, accurate and consistent to use molar mass in any bulk stoichiometric calculations. 
A mole of molecules most often contains a variety of molecular masses due to natural isotopes and the average is usually not identical to any single molecule. This is inconsistent with a strict interpretation of the definition because it neglects that the mass of a single molecule is not the same as the average of an ensemble. It is interpreted by many, including many chemists, to be a synonym of molar mass differing only in units (see average molecular mass below). Due to this relativity, the molecular mass of a substance is commonly referred to as the relative molecular mass, and abbreviated to Mr.Īlthough this term appears well-defined, there are varying interpretations of this definition. The molecular mass (abbreviated Mr) of a substance, formerly also called molecular weight and abbreviated as MW, is the mass of one molecule of that substance, relative to the unified atomic mass unit u (equal to 1/12 the mass of one atom of carbon-12). 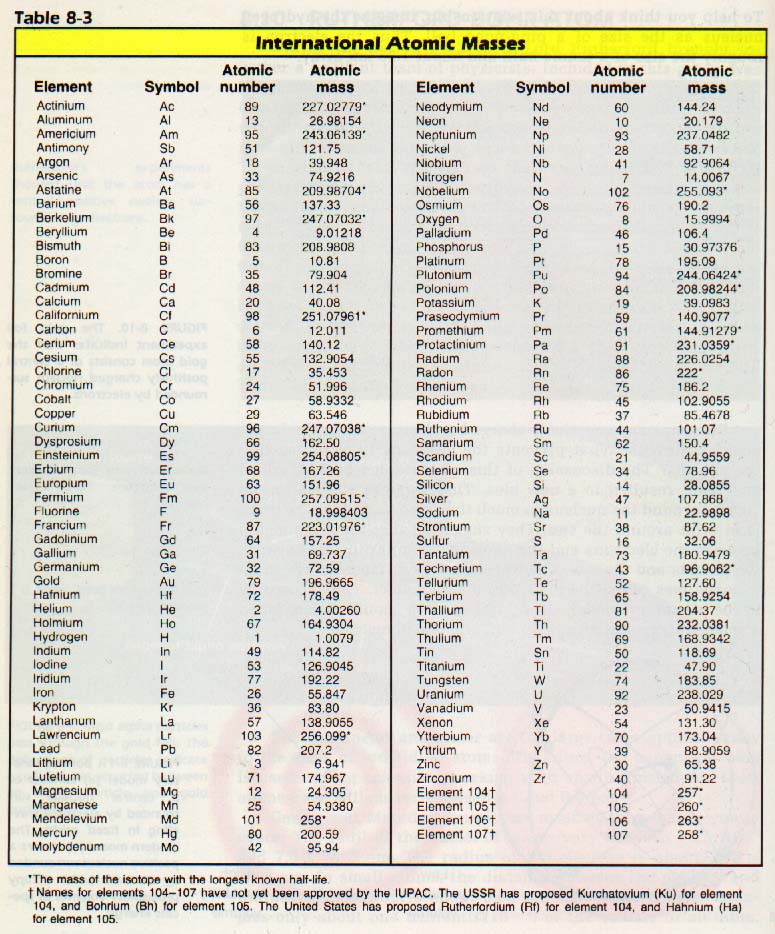
4 Example: Average Molecular Mass versus Molecular Mass versus Molar Mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
